The crystal structure of manganese makes it a hard and brittle metal, with low electrical and thermal conductivity. Below 727 ☌, a unit cell of 58 spatially diverse atoms represents the energetically lowest way of achieving a zero net magnetic moment. 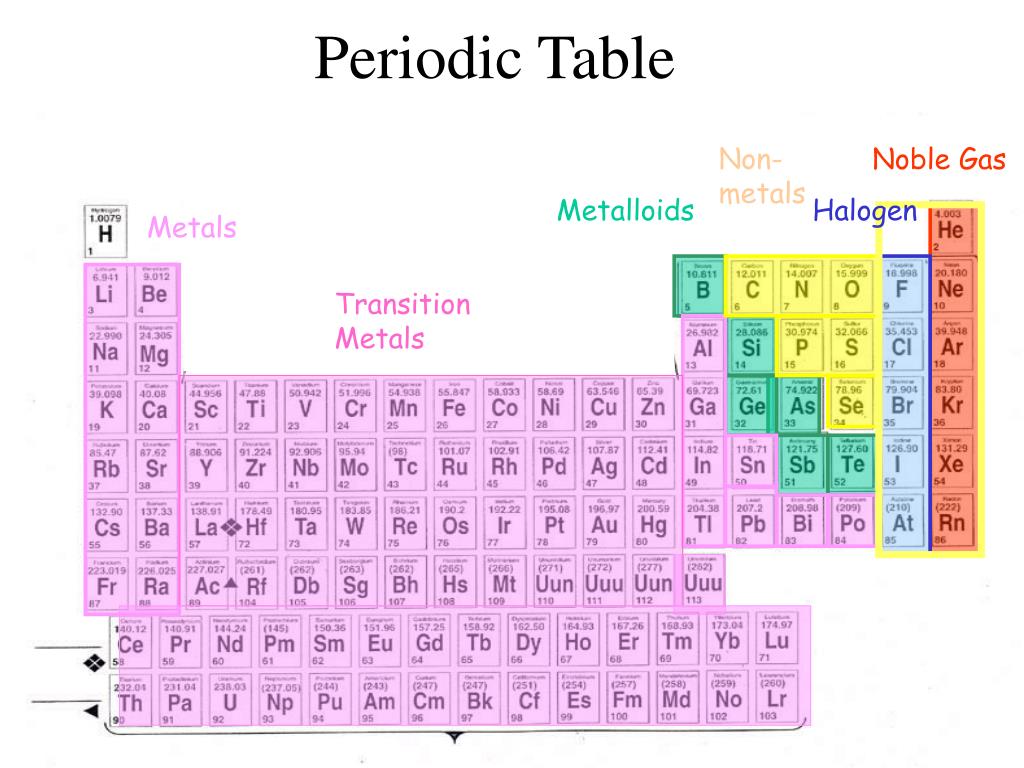
This confers a large magnetic moment on each atom. It has been described as resembling "a quaternary intermetallic compound with four Mn atom types bonding as if they were different elements." The half-filled 3d shell of manganese appears to be the cause of the complexity. Manganese has a complex crystal structure with a 58-atom unit cell, effectively four different atomic radii, and four different coordination numbers (10, 11, 12 and 16). Well-behaved metals have crystal structures featuring unit cells with up to four atoms.Several other aurides (KAu, RbAu) have since been synthesized, as well as the red transparent compound Cs 2Pt which was found to contain Cs + and Pt 2− ions. This was subsequently shown to consist of caesium cations (Cs +) and auride anions (Au −) although it was some years before this conclusion was accepted. In 1943, Sommer reported the preparation of the yellow transparent compound CsAu. Since then further compounds (" alkalides") containing anions of all other alkali metals except Li and Fr, as well as that of Ba, have been prepared. The synthesis of a crystalline salt of the sodium anion Na − was reported in 1974. The common notions that "alkali metal ions (group 1A) always have a +1 charge" and that "transition elements do not form anions" are textbook errors.Sodium, potassium, rubidium, caesium, barium, platinum, gold Within each category, elements can be found with one or two properties very different from the expected norm, or that are otherwise notable. Why, for example, was manganese such a bad conductor of electricity, when the elements on either side of it were reasonably good conductors? Why was strong magnetism confined to the iron metals? And yet these exceptions, I was somehow convinced, reflected special additional mechanisms at work. in the periodic table, anomalies too-some of them profound. Others count some of the metalloids as post-transition metals. Some authors count metalloids as nonmetals with weakly nonmetallic properties. Metalloids, straddling the metal-nonmetal border, are mostly distinct from either, but in a few properties resemble one or the other, as shown in the shading of the metalloid column below and summarized in the small table at the top of this section.Īuthors differ in where they divide metals from nonmetals and in whether they recognize an intermediate metalloid category. The characteristic properties of metals and nonmetals are quite distinct, as shown in the table below. Metalloids Tellurium, described by Dmitri Mendeleev as forming a transition between metals and nonmetals Specialized subcategories such as the refractory metals and the noble metals also exist. 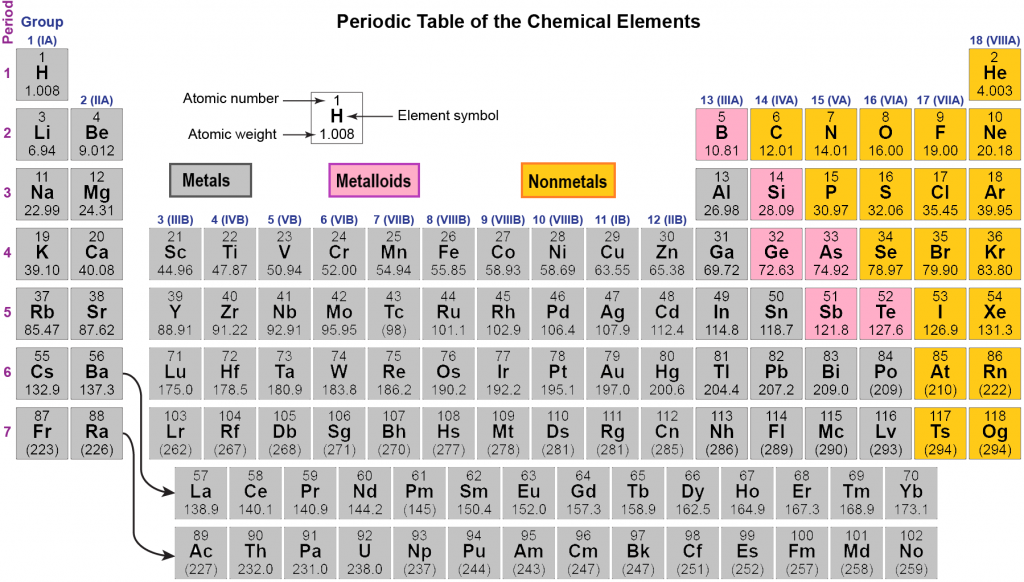
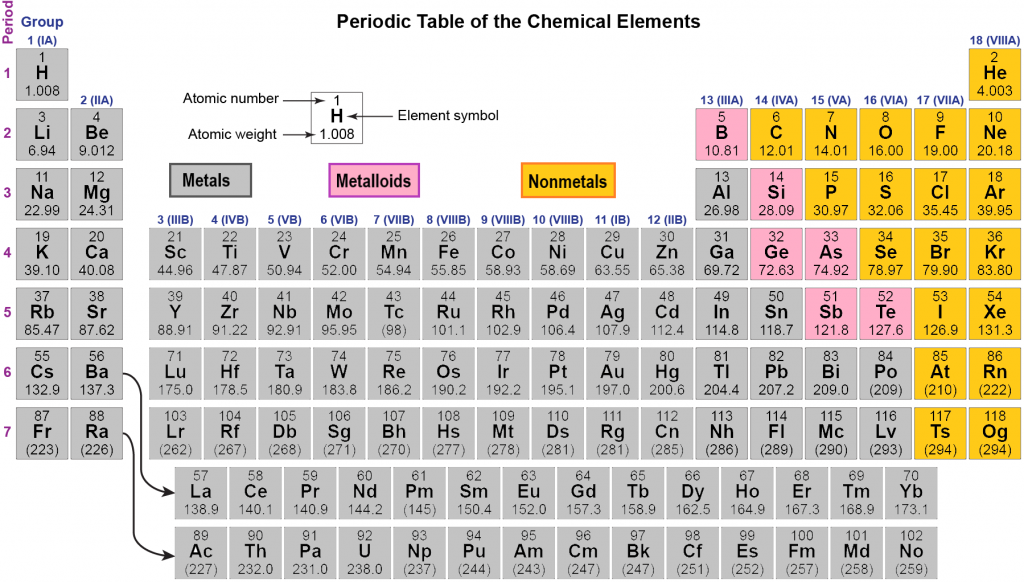
From left to right in the periodic table, these categories include the highly reactive alkali metals the less-reactive alkaline earth metals, lanthanides, and radioactive actinides the archetypal transition metals and the physically and chemically weak post-transition metals. Metals comprise the large majority of the elements, and can be subdivided into several different categories. Au, Pt), or have nonmetallic structures ( Mn and Ga are structurally analogous to, respectively, white P and I). Ti, Re), or are noble (hard to oxidise, e.g. W, Nb), are liquids at or near room temperature (e.g. Be, Al) or very high melting points (e.g. Some metals appear coloured ( Cu, Cs, Au), have low densities (e.g. Most metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and electronegativities, and are found naturally in combined states. Click on 'Element Atomic Number', 'Element Symbol', 'Element Name' and 'Element Color' headers to sort.Metals appear lustrous (beneath any patina) form mixtures ( alloys) when combined with other metals tend to lose or share electrons when they react with other substances and each forms at least one predominantly basic oxide. This Color table gives the Color of all the elements of periodic table. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
